|
However, his model worked well as an explanation for the emissions of the hydrogen atom, but was seriously limited when applied to other atoms. Moving up the ladder increases your potential energy, while moving down the ladder decreases your energy.īohr's work had a strong influence on our modern understanding of the inner workings of the atom. As you move up or down a ladder, you can only occupy specific rungs and cannot be in the spaces in between rungs. The Bohr model represents the structure of an atom developed by Danish physicist Niels Bohr in 1913. 
An everyday analogy to the Bohr model is the rungs of a ladder. The electron is not allowed to occupy any of the spaces in between the orbits. That’s all, this is our Bohr model of the Beryllium atom that contains 4 protons and 5 neutrons in the nucleus region, and 4 electrons are orbited around the nucleus, two electrons in the first shell, and two electrons in the second shell. The orbits that are further from the nucleus are all of successively greater energy. The ground state of the hydrogen atom, where its energy is lowest, is when the electron is in the orbit that is closest to the nucleus. When the electron is in one of these orbits, its energy is fixed. It accounts for a wide range of physical phenomena, including the existence of discrete packets of energy and matter, the uncertainty principle, and the exclusion principle.Īccording to the Bohr model, often referred to as a planetary model, the electrons encircle the nucleus of the atom in specific allowable paths called orbits. This is a theory based on the principle that matter and energy have the properties of both particles and waves. This was the basis for what later became known as quantum theory. When the energy is removed, the electrons return back to their ground state, emitting a corresponding amount of energy-a quantum of light, or photon. r We have seen that in Bohr’s model the coulombic force is assumed to be equal and opposite to the centrifugal force equation (1). 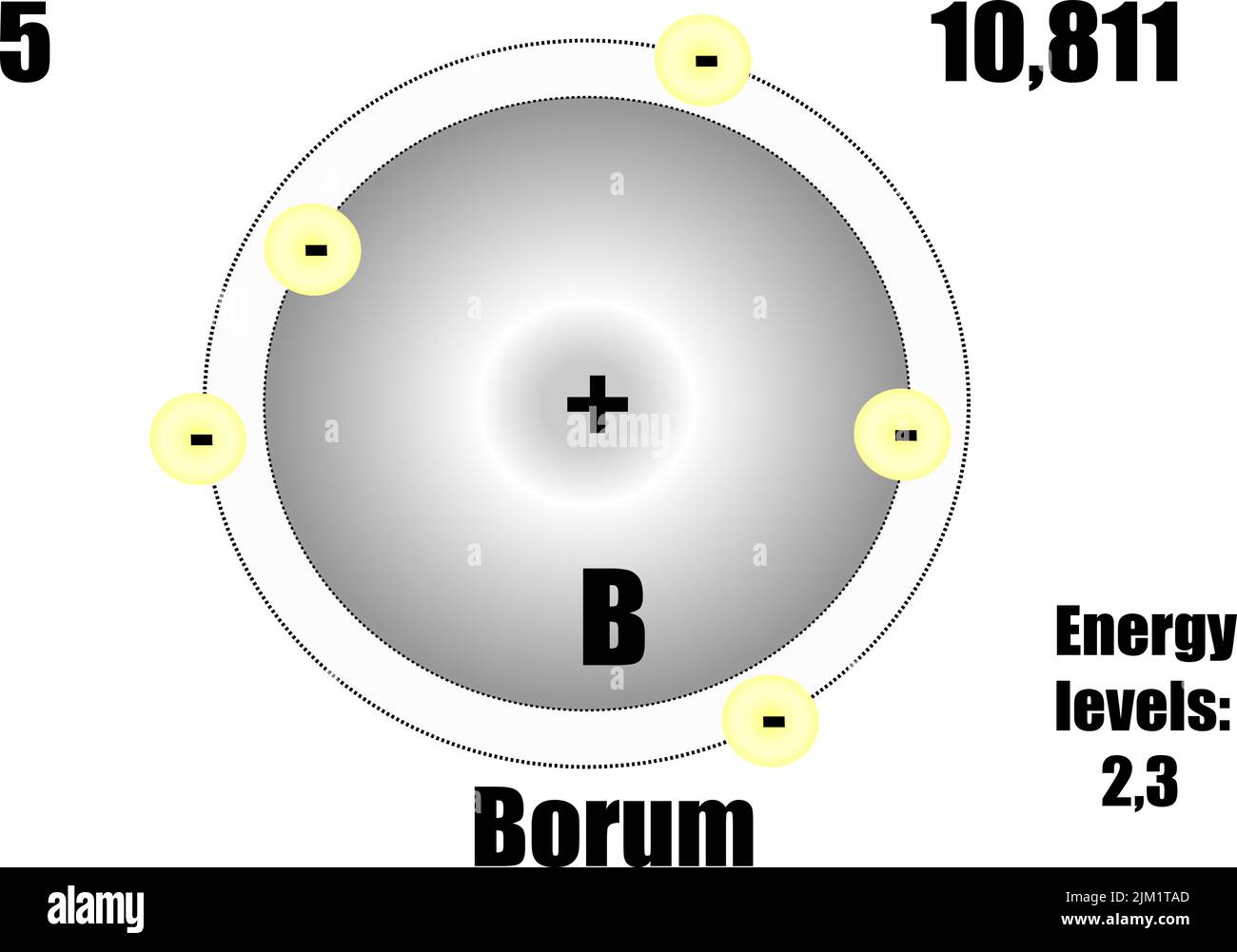
(Credit: Zachary Wilson Source: CK-12 Foundation License: CC BY-NC 3.0(opens in new window))īohr explained that electrons can be moved into different orbits with the addition of energy. The potential energy, which arises from the coulombic attraction between the negative charge of the electron and the positive charge in the nucleus, is given by U Ze2/r. \): Bohr's atomic model hydrogen emission spectra. SCIENCE The Bohr Model of Boron: An Overview The Bohr Model of Boron helps us understand the electron structure and its arrangement around the nucleus of boron atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
